AZF1 and AZF2 proteins regulate plant growth under drought and salt stresses
Description
Plants are exposed to various environmental stress conditions, such as drought, high salt, and low temperature. In response to these stresses, plants regulate growth and development by altering gene expression. Previous reports suggest that overexpression of some stress-inducible transcription factors can increase stress tolerance, resulting in growth inhibition. However, little is known about how such stresses cause plant growth inhibition. This study is aimed to elucidate the mechanism involved in plant growth regulation under environmental stresses by analyzing the functions of two Arabidopsis C2H2-type zinc-finger transcription factors (AZF1 and AZF2) that are induced by abiotic stresses.
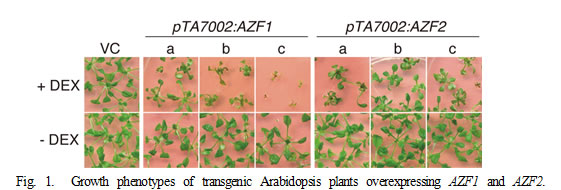
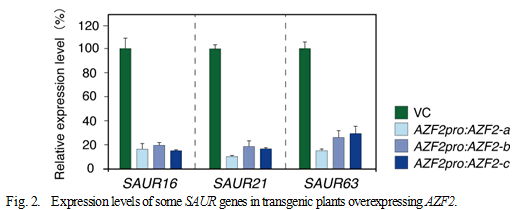
AZF1 and AZF2 genes encode C2H2-type zinc-finger proteins that are thought to function as transcriptional repressors. Expression of these genes are induced by osmotic stresses such as drought and high salt, and a phytohormone abscisic acid. Subcellular localization studies using GFP (green fluorescent protein) fusion proteins showed that the AZF1 and AZF2 proteins are localized to the nuclei in roots under control conditions, whereas the AZF2 protein accumulates in the nuclei of leaf cells under high-salinity stress. To analyze the functions of the AZF1 and AZF2 in plants, we generated transgenic Arabidopsis plants overexpressing AZF1 and AZF2 using stress-responsive promoters or a glucocorticoid-inducible promoter. These transgenic plants displayed dwarfed growth with smaller curled leaves (Fig. 1). It was also shown that overexpression of AZF1 and AZF2 enhances salt sensitivity in plants. Transcriptome analyses of the transgenic plants demonstrated that AZF1 and AZF2 repress the expression of various genes that are down-regulated by osmotic stresses and abscisic acid treatment. It is noteworthy that many “small auxin-up RNA (SAUR)” genes that may be involved in auxin-mediated cell elongation are down-regulated in the plants overexpressing AZF1 and AZF2 (Fig. 2).Moreover, gel mobility shift assays revealed that the AZF1 and AZF2 proteins directly interact with the SAUR promoter regions to repress the expression of these genes. Collectively, the results indicate that AZF1 and AZF2 function as transcriptional repressors which regulate plant growth under abiotic stress conditions.
The study’s findings suggest the possibility that the growth of plants can be artificially controlled under environmental stresses by adjusting the expression levels of some transcription factors including AZF1 and AZF2. Further studies are required to confirm the functions of proteins similar to AZF1 and AZF2 in crops, such as rice and soybean, to develop a technique promoting optimum plant growth under severe environmental conditions, thereby improving abiotic stress tolerance in crops and increasing crop yield.
Figure, table
-
pTA7002:AZF1, pTA7002:AZF2, and vector control plants were grown for two weeks on GM agar plates supplemented with or without 1 µM DEX.
-
The relative expression levels of the down-regulated genes were determined in the vector control and in three independent transgenic lines that were treated with 200 mM NaCl using qRT-PCR. The highest expression level of each gene was designated as 100.
- Affiliation
-
Japan International Research Center for Agricultural Sciences Biological Resources and Post-harvest Division
- Classification
-
Administration A
- Research project
- Program name
- Term of research
-
FY 2011 (FY 2011-FY 2015)
- Responsible researcher
-
Kodaira Shinsuke ( Biological Resources and Post-harvest Division )
KAKEN Researcher No.: 60649911Maruyama Kyounoshin ( Biological Resources and Post-harvest Division )
Fujita Yasunari ( Biological Resources and Post-harvest Division )
Shinozaki Kazuko ( University of Tokyo )
ORCID ID0000-0002-0249-8258 - ほか
- Publication, etc.
-
Kodaira et al. (2011) Plant Physiol 157, 742-756
- Japanese PDF
-
2011_08_A4_ja.pdf121.58 KB
- English PDF
-
2011_08_A4_en.pdf86.72 KB