Development of technology to determine maturity in important prawn species
Description
[Objectives]
In recent years, shrimp aquaculture has rapidly expanded and developed as an important commercial enterprise in Southeast Asian regions. However, in hatchery operations, lack of reliable methods for accurately determining maturation stage of spawners makes broodstock selection a difficult process, often leading to eggs and larvae of poor quality, and unstable production. Given this background, in order to enable selection of suitable female spawners for purposes of stable seed production, it has been necessary to develop a technology to determine ovarian maturity based on studies elucidating yolk protein structure and processing in economically important species of shrimp.
[Results]
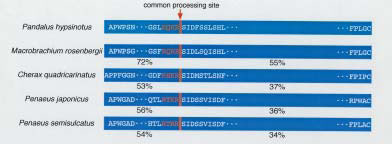
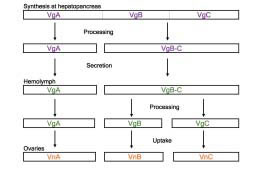
A full-length cDNA encoding vitellogenin (Vg) was firstly cloned and its complete amino acid sequence deduced in the giant freshwater prawn, Macrobrachium rosenbergii; subsequently, similar methods were employed to examine Vg in the kuruma prawn, Penaeus japonicus and the coonstriped shrimp, Pandalus hypsinotus. These results and those obtained by other authors thereafter have demonstrated that a high degree of identity exists among yolk proteins in various shrimp and prawn species (Fig. 1). Results obtained from electrophoretic and immunological analyses of the subunit composition of yolk proteins in the hemolymph and ovary in M. rosenbergii have clarified the site of synthesis and processing mechanisms of Vg. After being synthesized in the hepatopancreas as a precursor molecule, Vg is proteolytically cleaved into two subunits, VgA and VgB-C which are released into the hemolymph. In the hemolymph, the VgB-C subunit is further cleaved, forming two subunits VgB and VgC. The three subunits Vgs A, B and C are then sequestered by the ovary to give rise to Vn (Fig. 2). It is very likely that these processing mechanisms are common features among many prawn species. On the basis of the structural similarities of Vg in prawn species, an anti-serum against shrimp yolk protein has been developed and used in quantitative determination of hemolymph Vg levels in prawn and shrimp species by dot-blots and enzyme immunoassays. This thus-developed technology, "A process for determining maturing by using anti-serum against shrimp egg yolk protein," can therefore be used in the development of immunological kits to measure yolk protein levels. In combination with morphological observations, it will prove to be a useful and reliable tool for assessing maturity in aquaculture-important prawn species.
Figure, table
-
Fig. 1. Representation of vitellogenin primary structure in significant shrimp and prawn species. Degree of amino acid identity compared to Pandalus hypsinotus is indicated by percent. Location of a common processing site (see also Fig. 2) is indicated by the arrow. -
Fig. 2. Processing of vitellogenin (Vg) in Macrobrachium rosenbergii. Vg is initially produced at the hepatopancreas, undergoes processing at two different locations, and is taken into the ovaries as vitellin (Vn).
- Affiliation
-
Japan International Research Center for Agricultural Sciences Fisheries Division
- Classification
-
Technical A
- Term of research
-
FY2002 (FY2001-2003)
- Responsible researcher
-
WILDER Marcy Nicole ( Fisheries Division )
ORCID ID0000-0003-2114-2000KAKEN Researcher No.: 70360394JAYASANKAR Vidya ( Can Tho University )
JASMANI Safia ( Can Tho University )
TSUTSUI Naoaki ( University of Tokyo )
PHUONG Nguyen Thanh ( Can Tho University )
- ほか
- Publication, etc.
-
Wilder, M.N., Subramoniam, T. and Aida, K. (2002): Yolk proteins of Crustacea. In Reproductive Biology of Invertebrates. Adiyodi, K.G. and Adiyodi, R.G. (series eds), Volume XII - Recent Progress in Vitellogenesis. Raikhel, A.S. and Sappington, T.W. (eds). Science Publishers Inc., Enfield, NH, USA, 131-174.
Okuno, A., Yang, W-J., Jayasankar, V., Saido-Sakanaka, H., Huong, D. T. T., Jasmani, S., Atmomarsono, M., Subramoniam, T., Tsutsui, N., Ohira, T., Kawazoe, I., Aida, K. and Wilder, M.N. (2002): Deduced primary structure of vitellogenin in the giant freshwater prawn, Macrobrachium rosenbergii, and yolk processing during ovarian maturation. Journal of Experimental Zoology, 292, 417-429.
- Japanese PDF
-
2002_21_A3_ja.pdf975.54 KB
- English PDF
-
2002_21_A4_en.pdf62.47 KB