Improvement of drought stress tolerance by gene transfer of a transcription factor, AREB1, involved in ABA-responsive gene expression
Description
Due to the food crisis and environmental pollution, it is becoming ever more important to breed environmental stress-tolerant crops. Plant productivity is greatly affected by environmental stresses, such as drought and high salinity. Genetic engineering has high potential to improve the stress tolerance of crops using gene transfer technology. Several different approaches to improving the stress tolerance of plants by gene transfer have been attempted. In our strategy, a gene encoding a transcription factor involved in abscisic acid (ABA)-responsible gene expression was used.
The plant hormone ABA is produced under drought and high-salinity stress conditions and plays a pivotal role in tolerance to these stresses. Numerous drought- and high-salinity-stress-inducible genes have been reported in plants, and many of them are also activated by ABA. In analyses of the promoters of such ABA-regulated genes, a conserved cis-element designated ABA-responsive element (ABRE; PyACGTGGC), which controls ABA-regulated gene expression, has been identified. The drought-responsive expression of an Arabidopsis gene, rd29B, is mainly mediated by ABA. Two ABREs play a crucial role in the expression of rd29B as cis-elements. Using yeast one-hybrid screening, we cloned three different cDNAs encoding ABRE-binding proteins (AREB1, AREB2, and AREB3) of Arabidopsis. Expression of AREB1 and AREB2 is upregulated by ABA and drought and high-salinity stresses. Both AREB1 and AREB2 function as trans-acting activators, as identified by transient expression analysis in protoplasts.
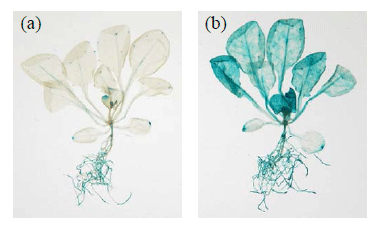
To determine the temporal and spatial expression patterns of AREB1, we analyzed transgenic Arabidopsis plants expressing an AREB1 promoter-GUS reporter gene. In unstressed plants, weak GUS expression was observed in the roots, leaf vascular tissues and hydathodes. By contrast, drought or ABA treatment of plants enhanced the AREB1 promoter activity in all tissues (Fig. 1). The subcellular localization of the AREB1 protein in plant cells was further analyzed using a Green Fluorescent Protein (GFP):AREB1 fusion protein. GFP fluorescence was detected in the nucleus, indicating that AREB1 is localized in the nucleus of the plant cells.
We generated transgenic plants overexpressing the AREB1 cDNA under the control of the CaMV 35S promoter (35S-AREB1). However, constitutive overexpression of intact AREB1 alone is not sufficient to activate its downstream genes such as RD29B under normal growth conditions. The ABA-induced modification of the AREB1 protein seems to be also required for the expression of its downstream genes. To overcome the masked transactivation activity of AREB1, we created an activated form of AREB1 (AREB1∆QT) carrying the AREB1 internal deletion mutants containing the bZIP DNA binding domain and transcriptional active domain of AREB1. We generated transgenic plants overexpressing AREB1∆QT and examined their stress tolerance. When plants grown in pots were not watered, almost all the wild-type plants died within 12 days. In contrast, nearly all the transgenic plants of two independent lines survived this level of drought stress and continued to grow when watering resumed, demonstrating their enhanced drought tolerance (Fig. 2). We analyzed upregulated genes in the transgenic plants using a microarray and found that eight genes in two groups were upregulated: LEA-class genes including RD29B and ABA- and drought-stress-inducible regulatory genes such as HIS1-3 (encoding a linker histone H1), GBF3 and RD20. All eight upregulated genes have at least two ABRE sequences in their promoter regions, and are known to be ABA- and stress-inducible. By contrast, an areb1 null mutant and a dominant loss-of-function mutant of AREB1 (AREB1:RD) with a repression domain exhibited ABA insensitivity. Further, AREB1:RD plants displayed reduced survival under dehydration. These data thus suggest that AREB1 regulates ABRE-dependent ABA-signaling that enhances drought tolerance in vegetative tissues. We believe that the activated form of AREB1 may be useful for improving the stress tolerance of agriculturally important crops by gene transfer.
Figure, table
-
Fig. 1. Histochemical localization of AREB1 promoter-driven GUS expression in Arabidopsis:
(a) 2-week old plant, (b) 2-week-old plant treated with 50 µM ABA. Bars = 5.0 mm -
Fig. 2. Enhanced tolerance to drought in the 35S-AREB1∆QT plants (2 independent transgenic plant lines).
Watering was withheld from 3-week-old plants for 12 days before the photograph was taken. Number codes = number of surviving plants out of the total.
- Affiliation
-
Japan International Research Center for Agricultural Sciences Biological Resources Division
- Classification
-
Technical A
- Term of research
-
FY2001~2005
- Responsible researcher
-
YAMAGUCHI-SHINOZAKI Kazuko ( Biological Resources Division )
FUJITA Yasunari ( Biological Resources Division )
FURIHATA Takashi ( Biological Resources Division )
SATO Rie ( Biological Resources Division )
NAKASHIMA Kazuo ( Biological Resources Division )
MARUYAMA Kyonoshin ( Biological Resources Division )
- ほか
- Publication, etc.
-
Fujita, Y., Fujita, M., Satoh, R., Maruyama, K., Parvez, M.M., Seki, M., Hiratsu, K., Ohme-Takagi, M., Shinozaki, K. and Yamaguchi-Shinozaki, K. (2005) AREB1 is a transcription activator of novel ABRE-dependent ABA-signaling that enhances drought stress tolerance in Arabidopsis. Plant Cell 17, 3470-3488.
Furihata, T., Maruyama, K., Fujita, Y., Umezawa, T., Yoshida, R., Shinozaki, K. and Yamaguchi-Shinozaki, K. (2006) ABA-dependent multisite phosphorylation regulates the activity of a transcription activator AREB1. Proc. Natl. Acad. Sci. U.S.A. 103, 1988-1993.
Yamaguchi-Shinozaki, K. et al. "Method for improving dessication stress resistance of plant by activated areb1" Japanese Patent Application No.2005-318871.
- Japanese PDF
-
2005_seikajouhou_A4_ja_Part3.pdf577.29 KB